Diagnostic Testing Solutions for Healthcare Providers
- Fast & Accurate
- Low Cost Options
- Track & Report Results
Order now and receive your diagnostic testing supplies within 24 hours.
A National Leader in Diagnostic Testing
GENETWORx recognizes that patients want to know more about their genetics and health in order to live healthier, more productive lives. Our scientists and researchers work tirelessly to advance Molecular Diagnostic Testing to help healthcare professionals diagnose and treat a broad range of conditions more quickly, safely and accurately.
Pathogen Testing Services
GENETWORx molecular pathogen testing identifies the specific pathogens that are causing the infection, as well as any antimicrobial resistance. The outcome is information that can help you select the optimal drug therapy for your patient.[1]
The PCR Testing Advantage
Detection
GENETWORx can detect more than 30 different types of pathogens and deliver 99% accurate results within 24-48 hours of lab sample receipt.
Accuracy
GENETWORx molecular pathogen testing is more accurate than standard culture testing. Our molecular pathogen testing detects anaerobic organisms growing deep inside that other tests may miss. [1]
Amplification
Speed
Our rapid turnaround time is within 24-28 hours from laboratory receipt.
Simplicity
Our simple to read report gives clinicians quick, actionable intelligence to make better treatment decisions.
Identification
Ability to identify pathogens when the patient is on empiric antibiotic treatment.
With molecular testing, not only can you receive a clear picture of the type and percent of pathogens causing the infection, in many cases, you can identify antimicrobial resistance. With the power of molecular testing, you can select the optimal medication therapy for your patient, improve treatment outcomes and reduce healthcare spend.
Discover Pharmacogenomics
- Pharmacogenomics is able to improve clinical outcomes as well as provide financial value to the health system. There are many compelling studies that found significant cost-savings value from introducing pharmacogenomics to physicians and patients. [3]
- GENETWORx is an expert in pharmacogenomics lab testing, detecting genetic differences that affect the metabolism of therapeutic medications and the possibility for adverse events.[3]
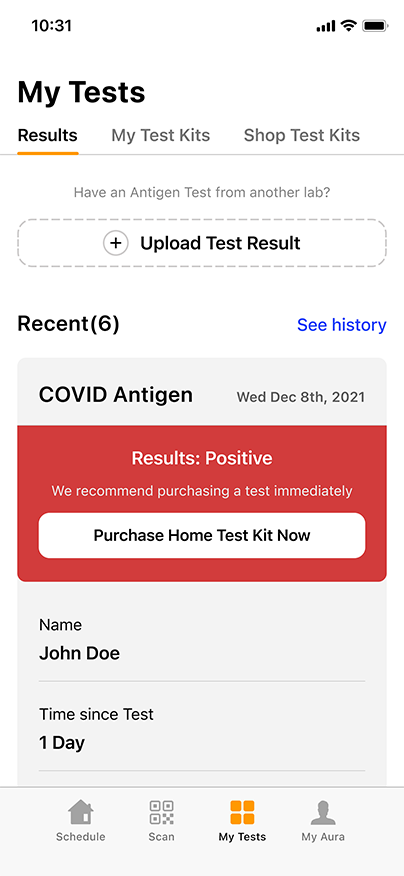
COVID-19 Testing Services
FLU A-B, COVID, RSV Testing for Your Patients
- COVID-19, influenza, and respiratory syncytial virus (RSV) are respiratory illnesses caused by different viruses. Because they have similar symptoms it can be difficult to tell them apart.
- GENETWORx is offering the Flu A-B/COVID Combo Test to detect both strands of influenza, RSV, and COVID-19 all from a single sample.
- With this combination test, healthcare providers can provide an accurate diagnosis to patients and treat them with the appropriate measures. Patients then can observe appropriate quarantine measures to stop the spread.
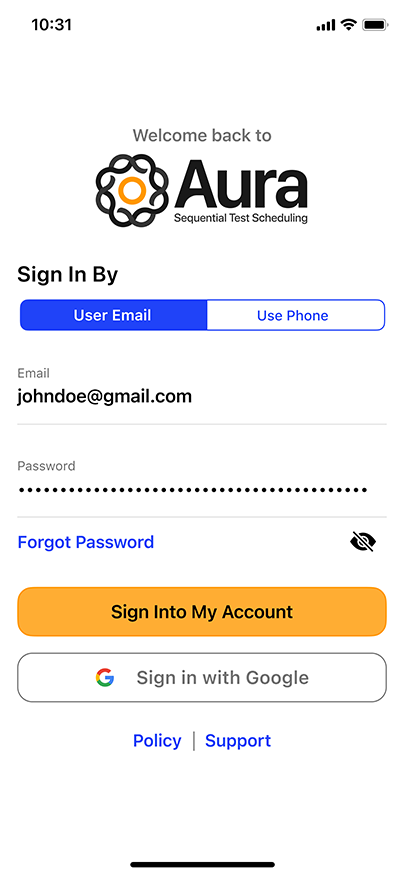
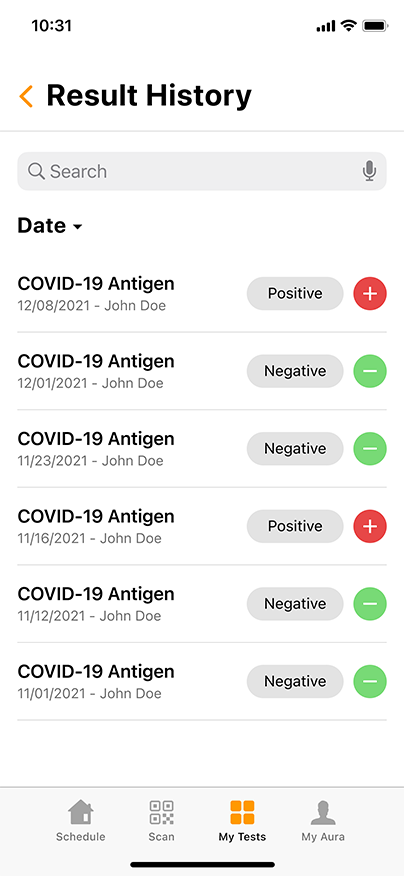
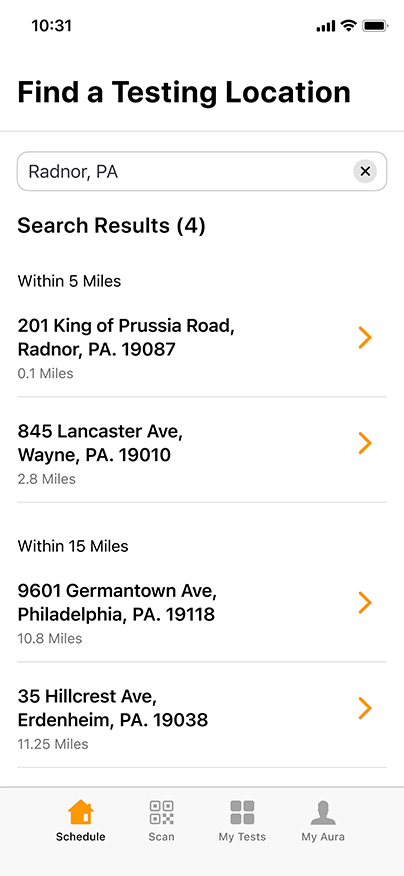
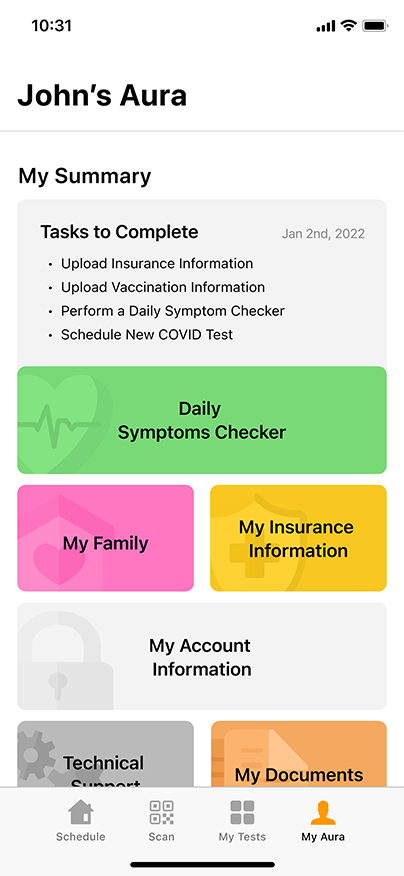
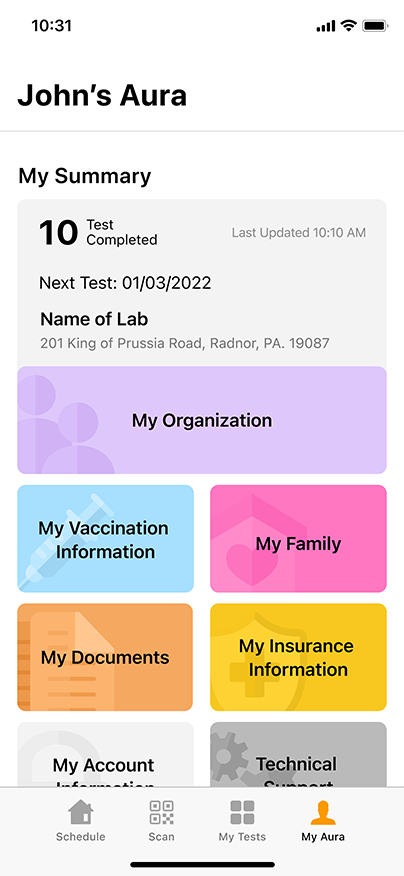
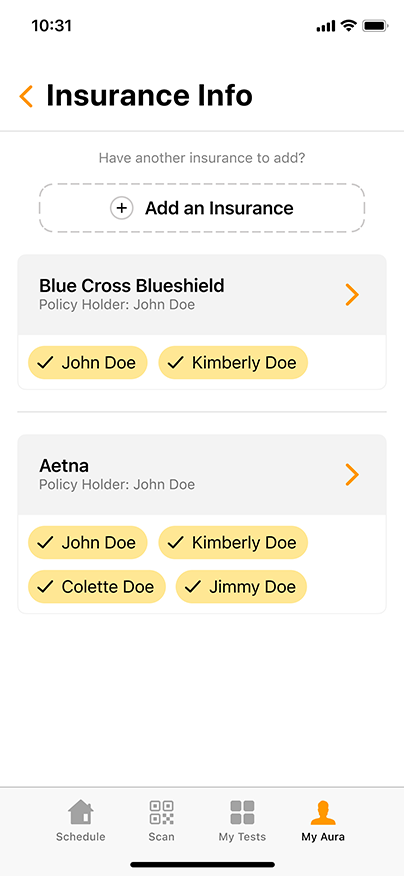
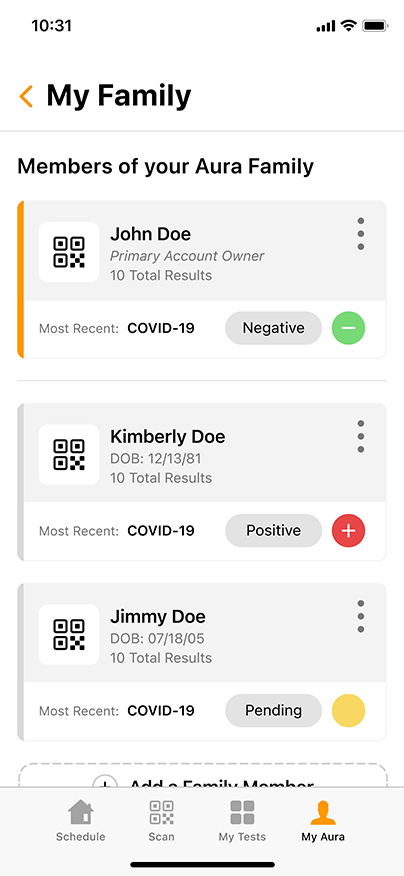
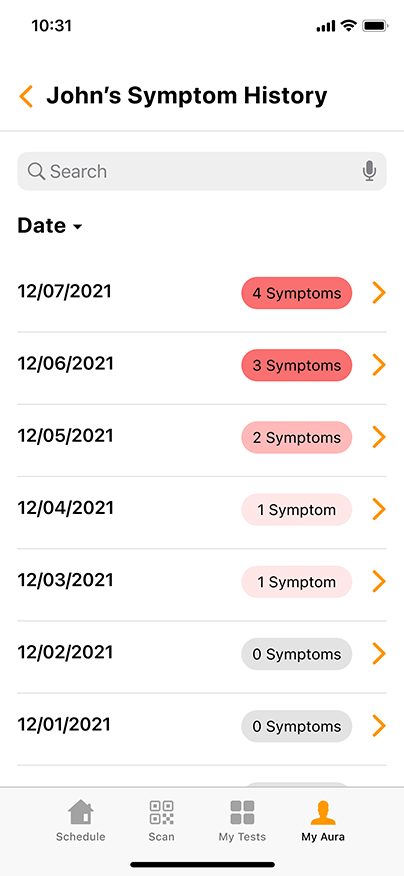
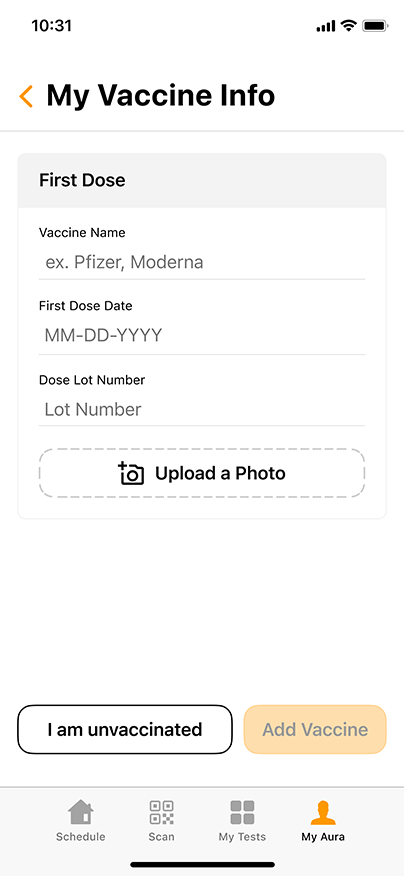
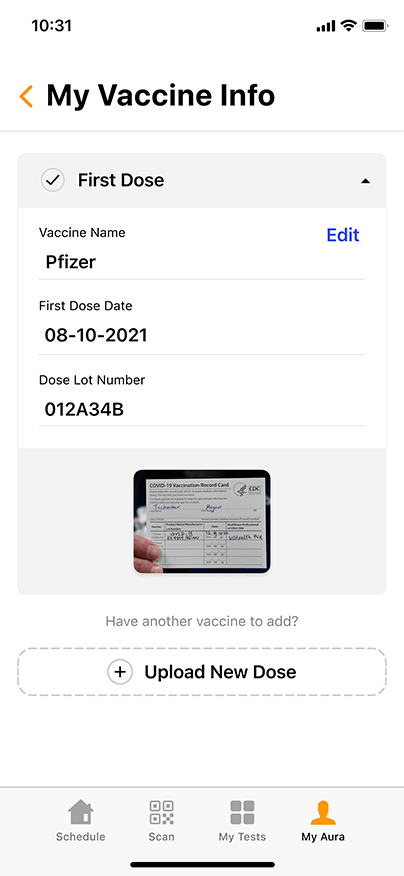
Convenient & Secure COVID-19 Digital Management
GENETWORx’s Aura software, provided by Nucleus, provides patients and customers a total end to end digital management and medical data system which provides:
- Test locations
- Test scheduling
- Sample tracking
- Daily symptom tracking
- Contact tracing assistance
- Dashboard compliance reporting
- Government reporting features
- Test Kit Activation (applies to the self-collection diagnostic COVID-19 Nasal Swab Test Kit)




Expansive Test Menu

Improving Patients Outcome