Employers
Testing
GENETWORx Is Empowering Businesses To Reopen, Stay Open, and Prosper During The Pandemic
COVID-19 Testing and Screening for Offices and Workplaces
The concerns over coronavirus have many employers and employees feeling uncomfortable with returning to the office, factory, store, or workplace. In fact, fears of COVID-19 infection and outbreak have forced many employees to quit their jobs and employers to shutter their businesses. Many economists believe fears over COVID-19 may actually put entire industries at risk of permanent and irreparable collapse.
There are businesses and business leaders who will rise above this current pandemic, and they will all have exceptional COVID-19 testing, screening, and security measures in place. Businesses across the country are partnering with GENETWORx to implement the world’s most accurate coronavirus testing and most comprehensive screening to ensure employees are comfortable returning to work, clients are comfortable resuming business, and customers are comfortable visiting the business.
GENETWORx provides on-site nasopharyngeal tests at your location of choice. GENETWORx also provides a Flu A-B/COVID Combo Test that detects two types of influenza viruses—influenza A and B—and differentiates them from RSV, and COVID-19. This combination test has a >96% accuracy rate for each virus.
GENETWORx is your one-stop partner in COVID-19 testing. In addition to, or instead of, providing on-site testing, GENETWORx can mail diagnostic COVID-19 self-collected nasal swab test kits to the homes of your employees. The GENETWORx COVID-19 Nasal Swab Test Kit received FDA emergency use authorization and is convenient, accurate and safe. With your order, employees receive a test kit to their address with the supplies and instructions needed to conduct a test, as well as the FedEx return mailer. Results are shared electronically with both employer and employee through an easy to use app, Aura, within 48 hours of lab receipt. Click to learn more about the self-collection COVID-19 Nasal Swab Test Kit.

Order Testing for Your
Business Today

GENETWORx Diagnostic Tests for COVID-19
GENETWORx provides accurate, reliable COVID-19 diagnostic tests. Every business partnering with GENETWORx is guaranteed the number of tests they need and tests conducted on-site will be reliably processed with results delivered less than 72 hours after the patient sample is collected. GENETWORx provides the nasopharyngeal tests for on-site collection, as well as a nasal swab test which can be mailed to the office or employees’ homes. All offer accuracy, convenience and peace of mind. Click to learn more about the self-collection COVID-19 Nasal Swab Test Kit option.
GENETWORx Processing for COVID-19
GENETWORx is performing COVID-19 testing using the same primer and probe sequences developed by the CDC in their Emergency Use Authorization to perform nucleic acid amplification and detection of COVID-19. At 99% specificity, our diagnostic tests are the most accurate in the world and are developed on-site at GENETWORx.
The primary processing workflow is performed by the below machines:
- Intelliqube instrument from LGC Douglas Scientific
- The Intelliqube is a combination liquid handler and real-time PCR instrument that can process up to 786 samples at a time
- CFX384 instrument from Bio-Rad
- The CFX384 is a rapid and sensitive real-time PCR instrument that can process 384 samples at a time
- Luminex Corporation’s ARIES SARS-CoV-2 Assay
- processes low-volume, rush accounts


GENETWORx Total Antibody Tests for COVID-19
The GENETWORx COVID-19 Total Antibody (IgM & IgG) assay is a blood test that is performed to determine if someone has been exposed/infected with the COVID-19 virus. The assay is 99.8% specific and 100% sensitive when performed at least 14 days after infection from COVID-19. If the person does not have symptoms but suspects they may have been infected, the antibody test can be performed at any time.
The antibody test checks to see whether a person’s immune system, the body’s defense against illness, has responded to the infection. It is not for diagnosing an active COVID-19 infection.
Total antibody tests detect both IgG and IgM in the blood to provide a clearer disease-state picture. These assays are more sensitive than the use of IgG or IgM alone for early detection of an immune response.
GENETWORx is More Than Just Testing
Testing alone does not create the peace of mind necessary to return to “business as usual.” Identifying carriers of COVID-19 is merely one step in creating a healthy and safe working environment. It’s what your business does after identifying cases of COVID-19 that truly matters.
GENETWORx is the manufacturer of the world’s most accurate COVID-19 test kits and the developer of the nation’s most comprehensive screening and security solution specifically designed for businesses, offices, factories, storefronts, buildings, and other workplaces.
Integrating with existing security systems, GENETWORx’s Sequential Testing for Population Management program ensures people entering your building recently and verifiably tested negative for COVID-19. This program arranges testing schedules, follows HIPAA guidelines for reporting, and organizes appropriate safety protocols in the event a COVID-19 carrier is identified on-site.
GENETWORx takes the guesswork out of local, state, and federal testing guidelines, security and safety protocols, and testing schedules. GENETWORx is a one-stop solution for total COVID-19 compliance.



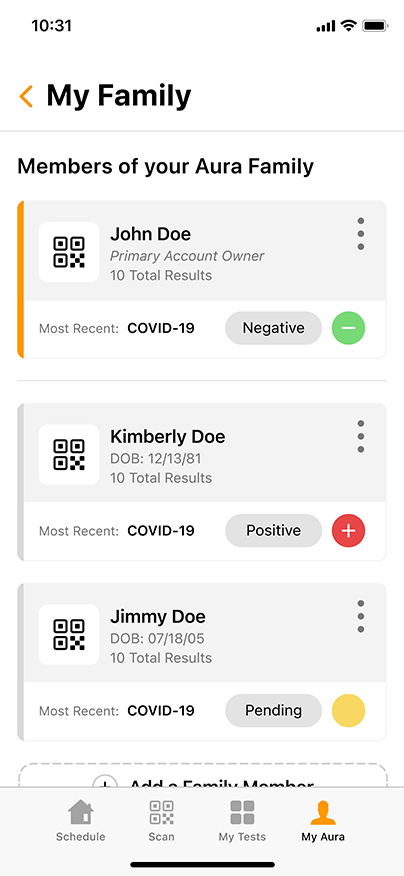
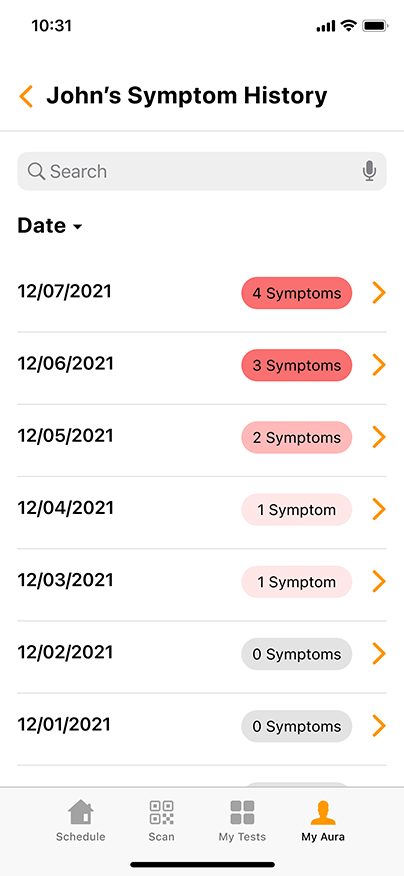
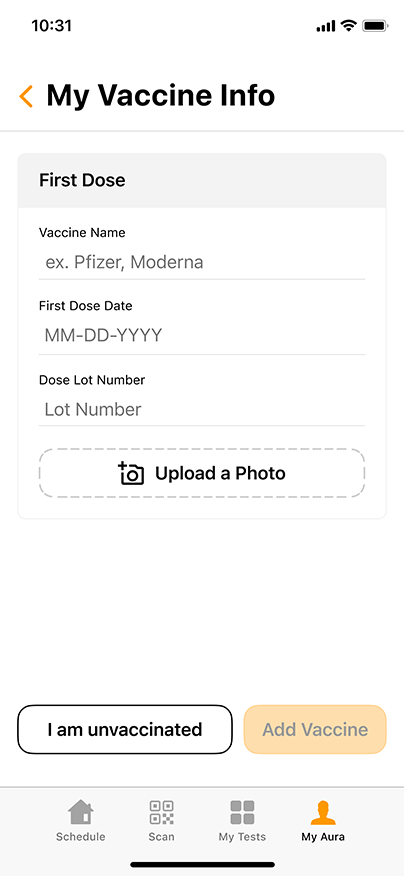
Aura: The COVID-19 Reporting and Security Software
Aura, the proprietary COVID-19 reporting and security software from GENETWORx, tracks sequential testing results and provides dashboards, accurate reporting, and important health data reflecting the COVID-19 status of your business.
Aura can easily integrate with your business’ security system, enabling you to screen for untested and coronavirus-positive persons at entry points to your workplace. Using standard QR-readers, Aura can cross reference individuals entering the workplace with their most recent COVID-19 test results, creating a discrete and effective way to suppress the presence of the contagion.
Aura is HIPAA compliant and can be customized to meet the needs of your specific business. GENETWORx can conduct independent assessments of your workplace and provide tailored security solutions to greatly mitigate the risks associated with COVID-19.
To learn more about Aura, click here.
Order Testing for Your
Business Today
GENETWORx Certified Status:
Giving Your Business The Competitive Edge
Business owners know that there are fewer competing businesses and more unemployed premium talent on the market right now than ever before. If you’re looking to grow your market share, you need to have a building that ensures uncompromised and unparalleled COVID-19 testing and security compliance.
Partnering with GENETWORx will give your business the competitive edge to transform this time of economic uncertainty into an opportunity to provide jobs and stimulate the marketplace when our country needs it most. GENETWORx will help custom-tailor a testing, screening, and security program for your business and workplace, creating the safest possible environment for your employees, staff, business partners and clients.
When you are GENETWORx Certified, your business will openly demonstrate its commitment to and dedication to keeping healthy. GENETWORx businesses will have an easier time recruiting top-tier talent, hosting meetings with major client accounts and business partners, and welcoming back customers.
GENETWORx is devoted to reactivating the American economy and will work with businesses throughout the country to implement the most sophisticated and comprehensive safety and security protocols. We are your greatest ally through this pandemic.

GENETWORx is the Preferred COVID-19 Testing Provider for:
How to Obtain On-site COVID-19 Diagnostic Test Kits for Your Organization

Contact GENETWORx
Contact GENETWORx. Within 72 hours, an account will be created and you will receive access to the GENETWORx portal for future test results.

Receive Test Kits
Within 24 hours the healthcare provider (HCP)/practice designated will receive a nasopharyngeal diagnostic test kits, or antibody testing supplies (if needed). The nasopharyngeal diagnostic test kit will include a nasopharyngeal swab, and the universal transport media in a vial to ensure RNA and DNA survival. Kits include a biohazard bag and postage paid, pre-addressed FedEx return pack.

Send Test Kits Back to GENETWORx
Once the test is administered (by the HCP for the nasopharyngeal test), the HCP or individual will put the vial and the test order with the ordering-physician’s signature in the provided bag and then into a prepaid FedEx package that will be delivered right to a GENETWORx lab.

Receive Results in Less Than 72 Hours
If the patient signed a release you will receive the results via the GENETWORx portal in less than 72 hours. Patients can also access their results directly from GENETWORx via phone, text or the patient portal. GENETWORx will report to the State Health Department, so you do not have to report anything to local health authorities.
GENETWORx also offers COVID-19 Nasal Swab Test Kits which may be self-administered and sent to your employees’ homes or locations of choice. Click to learn more about the self-collection COVID-19 Nasal Swab Test Kit option.
Enroll for the GENETWORx ‘Test Everyone USA’ Program
Businesses need to test 100% of their employees for COVID-19 in order to:
- Re-open business
- Restore a productive workforce and welcome employees back to work
- Meet customer demand for goods/services and generate revenue
- Ensure customer and employee confidence by creating a safe and healthy environment
- Provide the data to local, state and federal government, as well as medical experts to aid in research
- Stimulate the local and national economy
- Stop the spread of the COVID-19 virus
The ‘Test Everyone USA’ program, offered exclusively by GENETWORx, provides employers and organizations ongoing testing. Through this program, every employee will be tested for COVID-19 every 10 days, and receive test results in less than 72 hours. With this knowledge, everyone can have peace of mind and take the necessary actions based on their test results. Employers can focus on running their business with a productive, confident workforce and serve their customers and communities with the ‘COVID-19 Free, GENETWORx Certified’ seal.
Order Testing for Your
Business Today
COVID-19 Testing in The Workplace FAQs
Vaccines are considered the best weapon against deadly viruses, but vaccines are also not perfect. Similar to the flu, the CDC estimates that COVID-19 will mutate and evolve over time, creating a variety of strains. Therefore, the COVID-19 vaccine will display varying degrees of effectiveness based on how well the annual vaccine applies to each strain. Additionally, not every person in the country will get the vaccine which means coronavirus will likely see considerable rates of infection every year. Because there will likely never be a way to totally eradicate COVID-19, businesses need to safeguard against outbreaks which put staff at risk of prolonged illness. GENETWORx offers a testing and screening program which helps businesses keep employees safe and healthy. By sequentially testing every person inside a business, instances of COVID-19 will be identified quickly, allowing business owners to act immediately to suppress the spread and avoid disruptions to the business.
COVID-19 Diagnostic Testing FAQs
Physicians, hospitals, and any other United States healthcare provider can order GENETWORx’s 2019 Novel Coronavirus (COVID-19) test. Our tests are available for any patients who meet current guidance for evaluation of COVID-19 infection. Our nasopharyngeal tests feature 99% specificity and sensitivity to detect the presence of SAR-CoV-2, the virus responsible for COVID-19.
Call 610-726-1205 and a GENETWORx representative will set you up in GENETWORx’s portal and send the kits to your facility. Within 72 hours, your facility will be set up in our portal and within 24 hours your test kits will arrive.
After a physician contacts GENETWORx, we will send either the nasopharyngeal or saliva testing kits to your facility. The nasopharyngeal kit will include a swab and the universal transport media in a vial to ensure RNA and DNA survival. The saliva kit will include a specimen collection funnel, tube, and universal transport media and a cap for the tube. Once the swab is taken by a healthcare provider (for nasopharyngeal) or is self-administered (for saliva), the vial and test order with the physician’s signature are placed in an individual bag, which are then placed into a larger bag. This is sent via FEDEX to our laboratory, where it will be tested, and results will be sent through the GENETWORx portal within 2 to 3 business days.
GENETWORX’s nasopharyngeal swab collection is collected using a synthetic nasopharyngeal swab and placed in a viral transport medium to be accepted for testing. Only one sample type per patient will be accepted. Appropriately sized swabs with synthetic tips can be used with the exception of calcium alginate tips, swabs with preservatives, and swabs with wood shafts. Viral transport media that is acceptable for the collection of influenza specimens can be used to transport swabs for COVID-19 testing.
GENETWORX’s saliva specimen is gathered by the individual collecting their saliva into the funnel until the amount of liquid saliva reaches the 1 ml line. Only one sample type per patient will be accepted.
GENETWORx Laboratory provides both a nasopharyngeal test and a saliva test which can be conducted on-site. Both of these tests are extremely accurate: the nasopharyngeal test has a 99% sensitivity and specificity, and the saliva test yields an accuracy greater than 98%. GENETWORx also provides a self-collected COVID-19 Nasal Swab Test Kit which can be sent to the home of your employees. Click to learn more about the self-collection kit.
Once GENETWORX’s receives the provided sample, it will be tested and the result – whether it’s positive or negative – will be sent confidentially via our portal within less than 72 hours. You will receive instructions on how to set up the portal upon initial receipt of the COVID-19 test. Patients can also access their results directly from GENETWORx via phone, text or the patient portal.
In order to get quick, accurate results to patients, the following samples cannot be accepted for testing:
- Any swabs with calcium alginate, cotton tips, or wooden shafts
- Samples that have been refrigerated for more than 72 hours
- Samples that have been kept at room temperature for more than 24 hours
- Samples that are improperly labeled
- Samples that are contaminated
- Broken or leaking transport devices
For saliva tests, please note:
- You CANNOT eat, drink, smoke, or chew gum 30 minutes before providing your sample
- Do not remove the plastic film from the funnel lid
Genetworx is uniquely qualified to perform COVID-19 testing and deliver incredibly accurate, timely results because we regularly perform complex molecular diagnostic testing, outside of the pandemic. Not all companies have the technology and infrastructure to deliver an accurate result in a timely manner. We have special instrumentation and expertise to run a large volume of COVID-19 tests and produce results in less than 72 hours. Our nasopharyngeal test yields over 99% specificity and sensitivity and our saliva test has a greater than 98% accuracy. This capability is not found in most tests or in most laboratories. Some tests without this level of sensitivity will not detect the presence of the virus.
Depending on your state, GENETWORx is required by law to report positive and/or negative results to the state and public health departments.
GENETWORx’s is currently performing thousands of COVID-19 tests every day. We have been working closely with local and state officials to continue providing our services during this pandemic. Every day, GENETWORx is adding machinery automation and efficiencies to continually increase our testing capacity to meet the demand during this pandemic.
No, a physician does not have to be on staff. Rather, your company needs to partner with a physician. This way, the physician can order the tests, conduct the nasopharyngeal tests when they arrive, and work through GENETWORX’s portal to receive results. The saliva tests are self-administered, but still require a prescribing physician. If your company does not have a partnership with a physician, we can help you set one up so you are able to receive GENETWORx’s COVID-19 tests.
COVID-19 Antibody Testing FAQs
A venipuncture is performed to collect blood from the patient. Serum or plasma will be tested to determine if the patient has antibodies against COVID-19. Blood draws need to be performed by health care providers or at labs.
Results will be available in GENETWORx’s online portal or by phone (610-726-1205) within 24-48 hours after the sample has been received. If you provided your mobile number, you’ll receive a text from GENETWORx with a link to our secure portal.
GENETWORx COVID-19 Total Antibody (IgM & IgG) test is 99.8% specific and 100% sensitive when performed at least 14 days after COVID-19 infection.
Contact GENETWORx by calling 610-726-1205 to order your Total Antibody test. You’ll have a testing kit within 72 hours.
Yes. If you believe you’ve been exposed to COVID-19 or have tested positive but do not have symptoms, you can still get a GENETWORx Antibody test.
If you’ve tested positive for COVID-19, been at risk for exposure, have had symptoms or not, you are eligible for COVID-19 antibody testing.
There is no age minimum for antibody testing.
Although having the antibodies does not guarantee immunity, early research indicates there is at least some protection from previous COVID-19 exposure.
Because antibodies play such an important role in the battle against COVID-19, health care professionals need to know who has been exposed to COVID-19 and if they’ve developed an immune response. As we learn more and more about COVID-19, these results, combined with other important data, could help figure out individuals who are more (or less) susceptible to more COVID-19 infections.
If you test positive for COVID-19 antibodies, it likely means that you have been exposed to COVID-19. Antibodies are often the immune system’s way to fight off infections. However, antibodies don’t guarantee immunity against certain viruses. Unfortunately, we don’t know if people who have COVID-19 antibodies are protected in the future. Research is still ongoing.
Yes, you will want to social distance if you have COVID-19 antibodies. Just because you have antibodies doesn’t mean you’re immune from COVID-19 infection. Plus, you can transport the virus, increasing the risk of infection to other people.
No, you don’t have to get sick in order to get antibodies. Research shows that a large amount of people have the antibodies to COVID-19 without ever actually getting sick. That’s why some people test positive for COVID-19 without ever having any symptoms.
Simply having the presence of antibodies doesn’t mean you are immune against future COVID-19 infections. Antibody testing provides a yes or no when it comes to creation of antibodies to a specific virus.
Currently, there are no known medical risks associated with taking the COVID-10 Antibody test, beyond that of a typical blood draw.